The FDA said the recommendation was "out of an abundance of caution" as it investigates reports of six people developing "rare and severe" blood clots after receiving the vaccine.
ABC Action News is tracking the local impacts of the recommendation.
6:10 p.m.
Reporter JJ Burton is addressing concerns when it comes to the Johnson & Johnson COVID-19 vaccine
3:15 p.m.
The Agency for Health Care Administration is allowing those on Medicaid who were relying on the now-closed mobile sites to visit their website and find out what they need to do so they can get the vaccine.
11:45 a.m.
Publix says it suspended the program for the administration of the Johnson & Johnson vaccine.
10:30 a.m.
CVS Health is pausing the use of the J&J vaccine until further notice.
9:50 a.m.
Jeff Zients, White House COVID-19 Response Coordinator, released the following statement on the Johnson and Johnson vaccine.
As FDA and CDC announced earlier today, out of an abundance of caution, they have recommended a pause in the use of the Johnson & Johnson vaccine as they review data involving six reported U.S. cases of a rare and severe type of blood clot in individuals after receiving the Johnson & Johnson vaccine. The FDA and CDC will be providing details at their briefing later this morning.
This announcement will not have a significant impact on our vaccination plan: Johnson & Johnson vaccine makes up less than 5 percent of the recorded shots in arms in the United States to date. Based on actions taken by the President earlier this year, the United States has secured enough Pfizer and Moderna doses for 300 million Americans. Over the last few weeks, we have made available more than 25 million doses of Pfizer and Moderna each week, and in fact this week we will make available 28 million doses of these vaccines. This is more than enough supply to continue the current pace of vaccinations of 3 million shots per day, and meet the President’s goal of 200 million shots by his 100th day in office—and continue on to reach every adult who wants to be vaccinated. We are working now with our state and federal partners to get anyone scheduled for a J&J vaccine quickly rescheduled for a Pfizer or Moderna vaccine.
9:45 a.m.
Governor Ron DeSantis says Florida will follow the recommendation of the CDC and FDA by pausing the use of the Johnson and Johnson vaccines. DeSantis stressed that the recommendation is out of an abundance of caution and urged Floridians who already got the J&J shot without any adverse reaction not to worry.
"How many people are alive today because they had the J&J vaccine?" DeSantis said, adding, "There's no question it's saved lives already."
9:15 a.m.
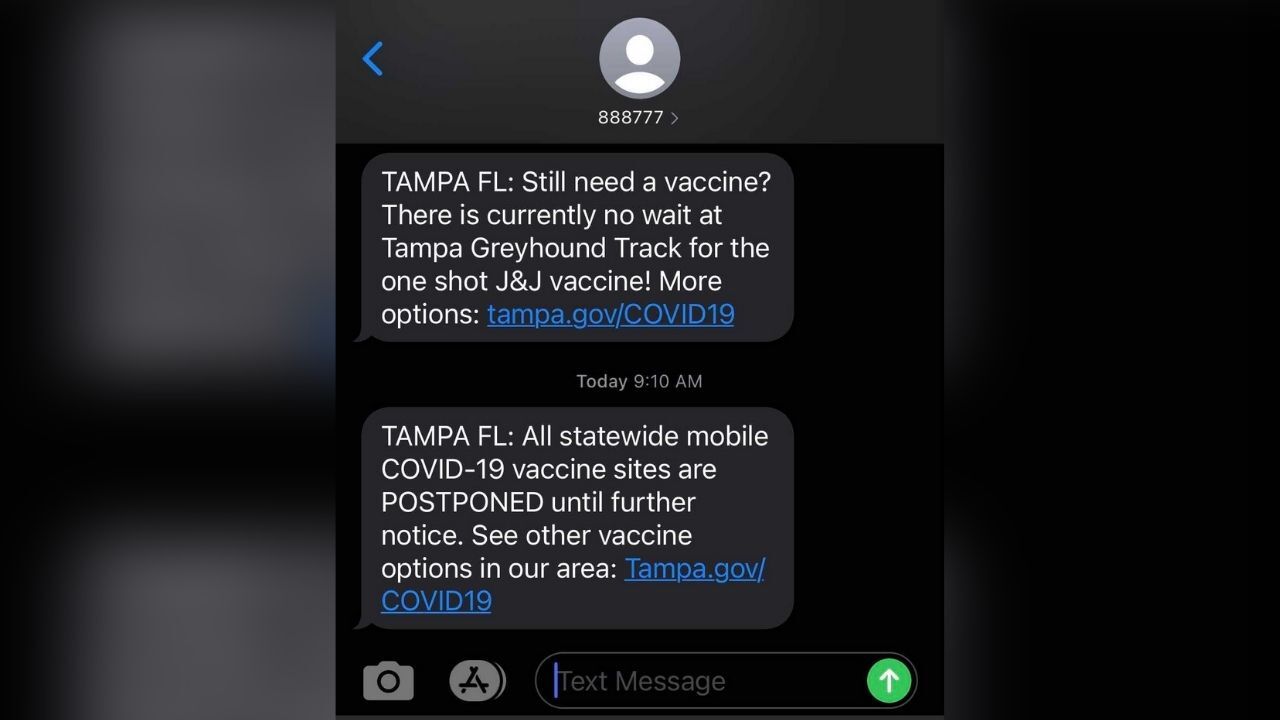
The federally-supported vaccine site at the Tampa Greyhound Track is no longer offering the Johnson & Johnson vaccine. At this time, the site is only administering the second dose of the Pfizer vaccine.
In light of the recent news from the @US_FDA and the @CDCgov, all J&J #COVID19 vaccine distribution has been paused. This also means the vaccine pop-ups have been paused and operations at the @femaregion4 Greyhound Track have been paused until further notice. #PublicHealth pic.twitter.com/X95SQvkHT5
— FL DOH in Hillsborough County (@DOHHillsborough) April 13, 2021
9 a.m.
All statewide mobile COVID-19 vaccine sites as postponed until further notice.